 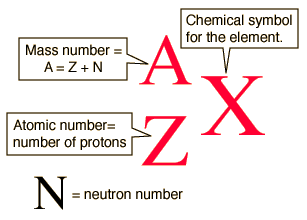
Examples of bis-silanes are bis-ethane, bis-tetrasulfane (bis-sulfur silane), and bis-amine (bis-amino silane). After condensation of SiOH groups, a cross-linked bis-silane film is expected to be denser than a cross-linked film prepared from a silane coupling agent. Assuming all OR groups hydrolyze for both silanes, the former would generate a maximum three silanol (SiOH) groups per molecule while the bis-silanes yield six SiOH groups per molecule. (RO) 3Si(CH 2) nR′(CH 2) nSi(OR) 3, where n = 0, 1, 2, 3 ….Ī major difference between bis-silanes and silane coupling agents listed in Table 1.1 is that silane coupling agents have one Si atom per molecule, attached by three hydrolyzable groups (OR), while bis-silanes have two Si atoms per molecule linked to six OR groups. 2.2.3 Other organofunctional trialkoxysilanes used in the corrosion control of metals Silanes react with inorganic surfaces to form metallo-siloxane covalent bonds for strong adhesion between silanes and inorganic substrates, while they react with polymers to form chemical bonds and IPNs for good silane/polymer adhesion. In brief, silane coupling agents perform as a “bridge” to promote adhesion between inorganic substrates (such as glass or oxide layers on metals) and polymers. Good adhesion of silanes to polymers is attributed to two mechanisms: (1) chemical reactions occurring between organofunctional groups in the silanes and reactive groups in the polymers and (2) the formation of interpenetrating networks (IPNs) at the silane/polymer interface. Silane bonding to polymers has also been studied. This improvement is because trialkoxysilane presents a more hydrophobic and highly cross-linked interphase region. Bonding of a trialkoxysilane R′Si(OH) 3 to silica has a much greater improvement in water resistance than a simple alkoxy bond between a hydroxyl functional polymer and silica. No direct evidence has been available so far for the equilibrium conditions at the interface, but the reversible nature of the siloxane-bonded interface has been demonstrated in performance testing. It is generally accepted that siloxane bonds formed at the silane/glass interface are hydrolyzable during long-term exposure to water and are reformable when dried. In the second step, the adsorbed silanol groups condense with surface hydroxyl groups to form siloxane (Si–O–Si) covalent bonds on glass or metallosiloxane (Me–O–Si) covalent bonds on a metal surface, releasing water. In the first step, silanol (Si–OH) groups from the hydrolyzed silanes adsorb to the inorganic substrate via hydrogen bonding to surface hydroxyl groups such as silanol (Si–OH) groups on a glass surface or Al hydroxyl (Al–OH) groups on an aluminum surface. Silane covalent bonding to an inorganic substrate, as shown in Fig. 1.2B, takes place via the following two steps. Therefore, determining the best balance between hydrolysis and condensation is one of the keys to the successful utilization of organosilane chemistry for special application. Important findings are as follows: (1) the rate of hydrolysis by both mechanisms is influenced by the nature of the organic group and the leaving alkoxy group attached to the silicon atom, and (2) pH also affects the hydrolysis rate, and the optimum pH for hydrolysis is not optimum for condensation. Mechanisms of acid- and base-catalyzed hydrolysis and condensation have been studied. Silane hydrolysis and condensation reactions, as shown in Fig. 1.2A, can be catalyzed by either an acid or a base. 
The process for organofunctional trialkoxysilane hydrolysis, condensation, and covalent bonding to an inorganic substrate (A) hydrolysis and condensation to form oligmers in the silane solution and (B) adsorption to an inorganic substrate (such as ceramics or surface oxide layers on metals) by hydrogen bonding and then covalent bonding to the substrate by a condensation reaction with hydroxyl groups. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
